A double-blind randomized trial of hyperbaric oxygen for persistent symptoms after brain injury
Patient characteristics and randomization
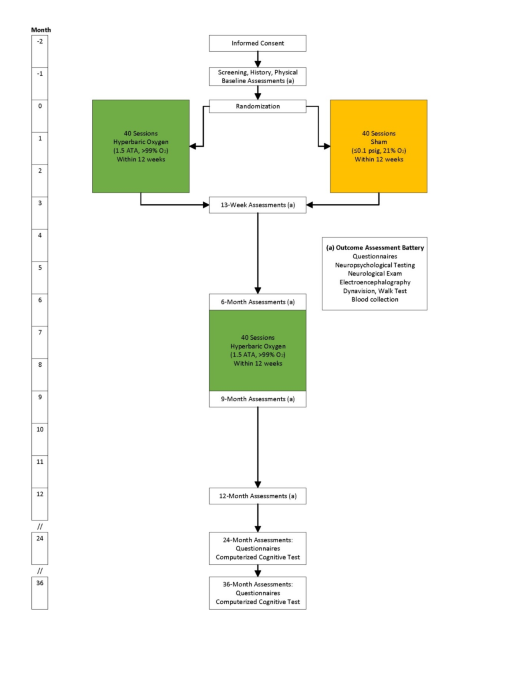
Participants were recruited from September 10, 2018 to April 28, 2021, when the trial was stopped due to exhausted funding and slowed recruitment. In that interval, 49 participants were randomized (26 to the HBO2 group, 23 to the sham group) (Fig. 2). After randomization, two sham participants were found ineligible and excluded; the final sample was 47 participants. There were no significant differences between groups in age, education, marital status, employment, race/ethnicity, and body mass index (Table 2). Estimated pre-morbid IQ was average or above for both groups (mean WASI-II Full Scale IQ, HBO2 111.2 ± 8.6, sham 119.6 ± 20.6). More sham participants used hypnotics or sedatives at baseline compared to the HBO2 group. Data at the prime outcome interval was analyzed on 42 participants (16 mild TBI, 4 severe TBI, 4 CO in HBO2; 11 mild TBI, 4 moderate TBI, 1 severe TBI, 1 CO, 1 hypoxic in sham) due to participant withdrawal of 5 participants (2 mild TBI in HBO2; 1 moderate TBI, 1 severe TBI, 1 CO in sham) prior to the 13-week assessments, and on 41 participants for the prime outcome measure due to 1 HBO2 participant with a mild TBI not being administered the NSI at the 13 week assessments due to tester error. Most participants (61.7%) suffered a single lifetime brain injury (HBO2 65.4%, sham 57.1%, p = 0.56). Four participants with a mild TBI qualifying injury had suffered a more severe TBI earlier in life (moderate TBI: 1 HBO2, 1 sham; severe TBI: 1 HBO2) or another type of brain injury (CO poisoning: 1 HBO2). Three participants in each group reported symptomatic head deceleration events (1 event: 2 HBO2, 3 sham; 3 events: 1 HBO2) in addition to their qualifying and lifetime brain injuries. One participant (HBO2 group) who had suffered CO poisoning received HBO2 treatment 1.3 years prior to randomization. Up to 5 other participants may have received prior HBO2 treatment, all over 6 years prior to randomization.
Study operations were interrupted due to the COVID-19 pandemic, affecting the study timeline of 17 participants (10 HBO2, 7 sham). Chamber sessions were paused for 9 participants (5 HBO2, 4 sham), while 5 participants’ scheduled chamber start dates were delayed (2 HBO2, 3 sham). Six participants had delayed outcome assessments (4 HBO2, 2 sham), and 4 had incomplete outcome assessments (3 HBO2, 1 sham). After study activities resumed, 3 participants (1 HBO2, 2 sham) did not continue their blinded chamber sessions. Forty-one participants completed the primary outcome assessment at 13 weeks. Outcome data was available for 41 participants at 6 months, 37 participants at 9 months, and 37 participants at 12 months (Fig. 2).

CONSORT diagram. Individuals screened, enrolled, randomized, and assessed throughout the study.
Prime outcome
At baseline, there were no significant differences in total NSI score between intervention groups. From baseline to 13 weeks, both groups’ total NSI scores decreased: HBO2 mean difference 10.6, 95% CI [6.0, 15.2], p < 0.001; sham mean difference 3.6, 95% CI [0.7, 6.5], p = 0.02. The HBO2 group reported a greater reduction in symptoms than the sham group (mean difference 7.0, 95% CI [1.7, 12.3], p = 0.01) and had decreased scores on all 3 NSI subdomains (cognitive, affective, and somatic), while only affective domain scores decreased in the sham group (Table 3).
NSI after 13 weeks
From baseline to 6 months, only the HBO2 group’s improvements were maintained, and the HBO2 group’s change score was greater than the sham group’s. Mean total NSI scores at 6 months were not significantly different between intervention groups (Table 4).
In the second chamber series, the HBO2 group experienced further improvements from 6 months to 12 months, but not from 6 months to 9 months (Table 4). The sham group’s scores did not change significantly during the second chamber series. From baseline to 12 months, the HBO2 group’s total change score was greater than that of the sham group, with improved scores in all 3 subdomains.
Only 2 eligible participants (1 HBO2, 1 sham) declined the 24-month assessments (11 HBO2, 7 sham completed); no eligible participants declined at 36 months (7 HBO2, 6 sham completed). From baseline to 24 months, only the HBO2 group’s total NSI score significantly improved. Both groups reported a significant decrease in symptoms from baseline to 36 months (Supplementary Table 1).
PTSD subgroup analyses
Individuals were included in the PTSD subgroup if their PCL-C score was ≥ 50 at baseline. Within the HBO2 group, the PTSD subgroup’s NSI change score was greater than that of the non-PTSD subgroup from baseline to 13 weeks (mean difference 12.9, 95% CI [4.5, 21.3], p = 0.004), and from baseline to 12 months (mean difference 12.1, 95% CI [1.8, 22.3], p = 0.02). There were no significant differences between PTSD vs. non-PTSD HBO2 subgroups from 6 to 9 months (mean difference 1.7, 95% CI [-13.7, 17.0], p = 0.82). Within the sham group, the PTSD subgroup’s NSI change score was greater than that of the non-PTSD subgroup from 6 to 9 months (mean difference 9.7, 95% CI [5.0, 14.4], p < 0.001), but not at other time points (baseline to 13 weeks mean difference 3.0, 95% CI [−3.6, 9.6], p = 0.34; baseline to 12 months mean difference − 4.6, 95% CI [−15.4, 6.1], p = 0.37).
Secondary outcomes
Full results are shown in Supplementary Tables 2–11. At baseline, the sham group performed better than the HBO2 group on both the short delay and long delay subtests of the California Verbal Learning Test (CVLT-II), but not on immediate recall nor on percentage of words retained (short delay free recall v Trial 5 z-score mean difference = 0.4, 95% CI [−0.1, 0.9], p = 0.09). On the Alcohol Use Disorders Identification Test-Consumption (AUDIT-C), 5 HBO2 participants reported potentially problematic alcohol use, compared to 1 sham participant, but that difference did not reach statistical significance. At 6 months, the sham group performed better than the HBO2 group on the code substitution delayed subtest of the ANAM, but there were no significant differences on the code substitution learning subtest of the ANAM, in which participants are asked to initially encode information. The HBO2 group had higher accuracy on the UPSIT compared to sham according to raw score, but not the normed percentile (p = 0.07). Otherwise, there were no significant differences between the groups at baseline and 6 months.
Self-report questionnaires other than the NSI
The PGIC was analyzed as a continuous variable as a research measure. At 13 weeks, PGIC scores did not differ significantly between groups, and PGIC scores increased (improved) in both groups between 13 weeks and 12 months. From 6 to 9 months, the sham group reported significantly greater improvement than the HBO2 group. At 12 months, the mean PGIC score for the HBO2 group exceeded 5, the threshold for significant, favorable change in a clinical setting.
Only 13% of participants reported moderate or severe anxiety symptoms on the Beck Anxiety Inventory (BAI) at baseline. At 13 weeks, only the HBO2 group’s BAI scores significantly improved (mean difference 3.0, 95% CI [0.7, 5.2], p = 0.01). From 6 to 9 months, neither intervention groups’ scores changed significantly. The HBO2 group reported a greater improvement in BAI scores compared to the sham group from baseline to 12 months (mean difference 4.5, 95% CI [0.6, 8.4], p = 0.03).
At baseline, 25.5% of participants’ Centers for Epidemiological Studies-Depression Scale (CES-D) scores suggested moderate depression and 44.7% suggested severe depression. From baseline to 13 weeks, both groups’ CES-D scores improved. This improvement remained significant within the HBO2 group at 6 months, but not in the sham group. From 6 to 9 months, neither intervention groups’ scores changed significantly, and from baseline to 12 months, both groups’ scores improved.
From baseline to 13 weeks, both groups’ Post-Traumatic Stress Disorder Checklist-Civilian Version (PCL-C) scores improved. Neither intervention groups’ scores changed significantly from 6 to 9 months. From baseline to 12 months, only the HBO2 group’s improvement was significant.
Both groups’ headache impact test (HIT-6) scores improved from baseline to 13 weeks, and from baseline to 12 months. There was no difference by intervention. Neither group’s scores significantly changed from 6 to 9 months. Only the HBO2 group’s Pittsburgh Sleep Quality Index (PSQI) scores improved from baseline to 13 weeks, and from baseline to 12 months. Neither group’s scores significantly changed from 6 to 9 months. On the vestibular symptom questionnaire (VSQ), the number of items endorsed as abnormal decreased in the HBO2 group from baseline to 13 weeks. Changes at other time points did not reach statistical significance.
On the Glasgow Outcome Scale (GOS-E), the sham group’s scores improved from 6 to 9 months. Changes at other time points did not reach statistical significance. From baseline to 13 weeks, both groups’ total Mayo-Portland Adaptability Inventory (MPAI-4) scores improved. Both groups’ improvements were maintained through 12 months, with the HBO2 group’s change score from baseline to 12 months greater than sham. Neither group’s scores significantly changed from 6 to 9 months.
From baseline to 13 weeks, both groups’ Rivermead Post-Concussion Questionnaire (RPQ) scores improved. From 6 to 9 months, only the sham group’s score improved significantly. From baseline to 12 months, scores improved in both groups with the HBO2 group reporting a greater improvement than the sham group.
On the World Health Organization Quality of Life (WHOQOL-BREF), both groups’ physical health domain scores improved from baseline to 13 weeks, and from baseline to 12 months. In both the psychological and environmental domains, neither group’s scores changed significantly from baseline to 13 weeks; only the HBO2 group reported significant improvement from baseline to 12 months. In the social domain, neither group’s scores significantly changed at any point. From 6 to 9 months, no significant changes occurred in any domain.
Neuropsychological testing
Embedded and stand-alone measures of effort indicated that results of neuropsychological testing were valid estimates of current cognitive functioning. From baseline to 13 weeks, the HBO2 group improved on 7 measures, while the sham group improved on 4. From 6 to 9 months, only the HBO2 group improved (2 measures). In addition, immediate recall on CVLT-II improved in the HBO2 group compared to the sham group from 6 to 9 months. From baseline to 12 months, the HBO2 group improved on 8 measures, while the sham group improved on 10. On category fluency, performance declined from baseline to 13 weeks in the sham group, and from 6 to 9 months in the HBO2 group, possibly related to use of alternate forms. On BVMT-R, performance declined from 6 to 9 months in the sham group, but their scores returned to baseline by 12 months.
Computerized neurocognitive testing
Both groups’ composite ANAM scores improved from baseline to 13 weeks and from baseline to 12 months. From 6 to 9 months, the HBO2 group improved on simple reaction time; no changes in the sham group reached significance.
Neurological tests
The baseline neurological examination was abnormal in 93.6% of participants, most commonly the Sharpened Romberg64 time (Supplementary Tables 8–9). There were no consistent improvements in either intervention group.
On Dynavision, both groups improved on forced choice target hits and reaction time from baseline to 13 weeks. Only the HBO2 group increased self-paced target hits. From 6 to 9 months, the HBO2 group improved on self-paced target hits and reaction time. From baseline to 12 months, both the HBO2 and sham groups improved on forced choice target hits and reaction time, and self-paced target hits. In addition, the HBO2 group improved on self-paced reaction time.
On a performance-based test of olfaction normed for age and gender, the majority of participants’ scores were normal at baseline (HBO2 = 57.7%, sham = 61.9%, p = 0.77). Only the HBO2 group demonstrated significant improvement from baseline to 13 weeks. By 12 months, average accuracy regressed to baseline performance. The sham group significantly declined from baseline to 6 months, but their scores returned back to baseline by 12 months.
Changes in distance walked on the 6MWT test did not reach significance across any time point comparisons for both groups. Both the neurological examination and 6-Minute Walk Test were insensitive to measuring change in this study.
Of 49 baseline EEGs, 10 were abnormal (6 HBO2, 4 sham). Of 5 participants in the HBO2 group with a baseline EEG and at least 1 subsequent EEG, 1 participant improved by 13 weeks, 1 by 6 months, and 1 by 9 months. In the sham group at 13 weeks or 6 months, 2 participants improved and 2 were unchanged from baseline. Of 3 participants in the sham group who had an abnormal baseline EEG and a post-HBO2 EEG, 1 improved and 2 were normal at 9–12 months. Across all participants who received HBO2 either initially or after 6 months, 6 of 8 improved or normalized. (Supplementary Table 12).
Blinding
Blinding of group allocation was successful at 13 weeks. Following the blinded intervention (at the 13-week assessment), 34.8% (n = 8) of participants in the HBO2 group and 23.5% (n = 4) in the sham group correctly guessed their group assignment, while 65.2% (n = 15) in HBO2 and 76.5% (n = 13) in sham guessed incorrectly or responded “not sure.” Two participants (1 HBO2, 1 sham) did not receive the questionnaire due to tester error. There was no significant difference in the odds of guessing randomization assignment correctly between groups (p = 0.55, OR 1.7, 95% CI [0.4, 7.1]), but participants were more likely to guess their randomization assignment incorrectly or respond “not sure” than to guess correctly (p = 0.01). At 9 months, after all participants had received HBO2, more participants correctly selected their original allocation, but were not more likely to select correctly than chance (p = 0.33). There were no between-group differences in accuracy (correct: 50% (n = 11) HBO2, 68.8% (n = 11) sham; incorrect or “not sure”: 50% (n = 11) HBO2, 31.3% (n = 5) sham, p = 0.24, OR 0.4, 95% CI [0.1, 1.8]).
Adverse events
From baseline through the 12-month assessment, chamber-related adverse events were reported by 19 of the 49 participants who were randomized (39%) (Supplementary Table 13). Three participants had adverse events related to study procedures other than the chamber sessions: a bruise after bumping into study equipment (n = 1), breach of confidentiality (n = 1), and syncopal episode during blood draw (n = 1). No serious adverse events were reported in this time frame. One participant with a pre-existing history of a mood disorder was discontinued in the multiplace sessions after reporting a worsening of symptoms during both the sham and intervention sessions, and subsequently withdrew from the study due to time/ inconvenience. One participant elected to discontinue chamber sessions due to concern of contracting SARS-CoV-2 in the clinic environment and confidentiality concerns but remained enrolled in the study.
During study participation, 5 participants (2 HBO2, 3 sham) reported one additional mild TBI. One additional HBO2 participant reported 2 mild TBIs. The injuries were reported at the 13-week (1 HBO2, 1 sham), 6-month (1 HBO2), 9-month (1 sham), 12-month (2 HBO2), and 36-month (1 sham) assessments. Two sham group participants reported a symptomatic head deceleration event not meeting mild TBI criteria (1 at 12 and 24 months, 1 at 36 months), and 1 HBO2 group participant reported such an event at the 6-month assessment, followed by negative health effects from environmental toxins at the 9-month assessment. Four participants reported unresolved sequelae of a COVID-19 infection67 at the 9-month (1 HBO2, increased dizziness and microsmia), 12-month (1 HBO2, cardiovascular and autonomic abnormalities, myalgia, and cognitive difficulties) and 36-month assessments (2 sham, microsmia, arthralgias/myalgias, fatigue, headaches, dizziness and nausea, and cognitive difficulties). When controlling for randomization group, experiencing additional brain injuries through the 12-month assessments did not influence patient-reported change score on the NSI from baseline to 12 months (estimate − 1.7, 95% CI [-10.3, 6.9], p = 0.69).
Protocol adherence
Participants who did not complete all 80 chamber sessions fell into two categories: Those who withdrew altogether (4 HBO2, 15%; 5 sham, 24%) and those who remained enrolled in the study but did not complete all chamber sessions (1 HBO2, 4%; 3 sham, 14%) (Fig. 2). All HBO2 group participants who did not complete all 80 chamber sessions had a mild TBI as their qualifying injury and were employed at baseline. In comparison, sham group participants who did not complete all 80 chamber sessions had a more severe qualifying TBI (3 moderate, 2 severe) or CO poisoning (1) and half (4) were on disability.
Per-protocol analysis
See Supplementary Tables 14–15. The only difference between the intention-to-treat analyses and the per-protocol analyses were within the baseline to 9-month assessments. In the intention-to-treat analysis for this interval, the mean difference in the HBO2 group was 8.4 (95% CI [4.5, 12.2], p < 0.001) and in the sham group was 4.2 (95% CI [0.6, 7.8], p = 0.02). The between-group mean difference at this timepoint was 4.2 (95% CI [-1.0, 9.4], p = 0.11). In the per-protocol analysis for this interval, the mean change score in the sham group was not statistically significant.
Qualitative results
Of 37 participants who completed the exit interview at 12 months, 34 (91.9%) participants endorsed ≥ 1 benefit from study participation, and 3 participants (all HBO2) did not. The most frequently cited benefits were improvements to overall quality of life (72.7% HBO2, 53.3% sham), cognition (54.5% HBO2, 60% sham), and mood (45.5% HBO2, 40% sham) (Supplementary Table 16).
link